NEURAL EXPLANATIONS FOR ANOREXIA
Biological explanations for anorexia nervosa, including neural explanations
NEURAL MECHANISMS INVOLVED IN ANOREXIA NERVOSA
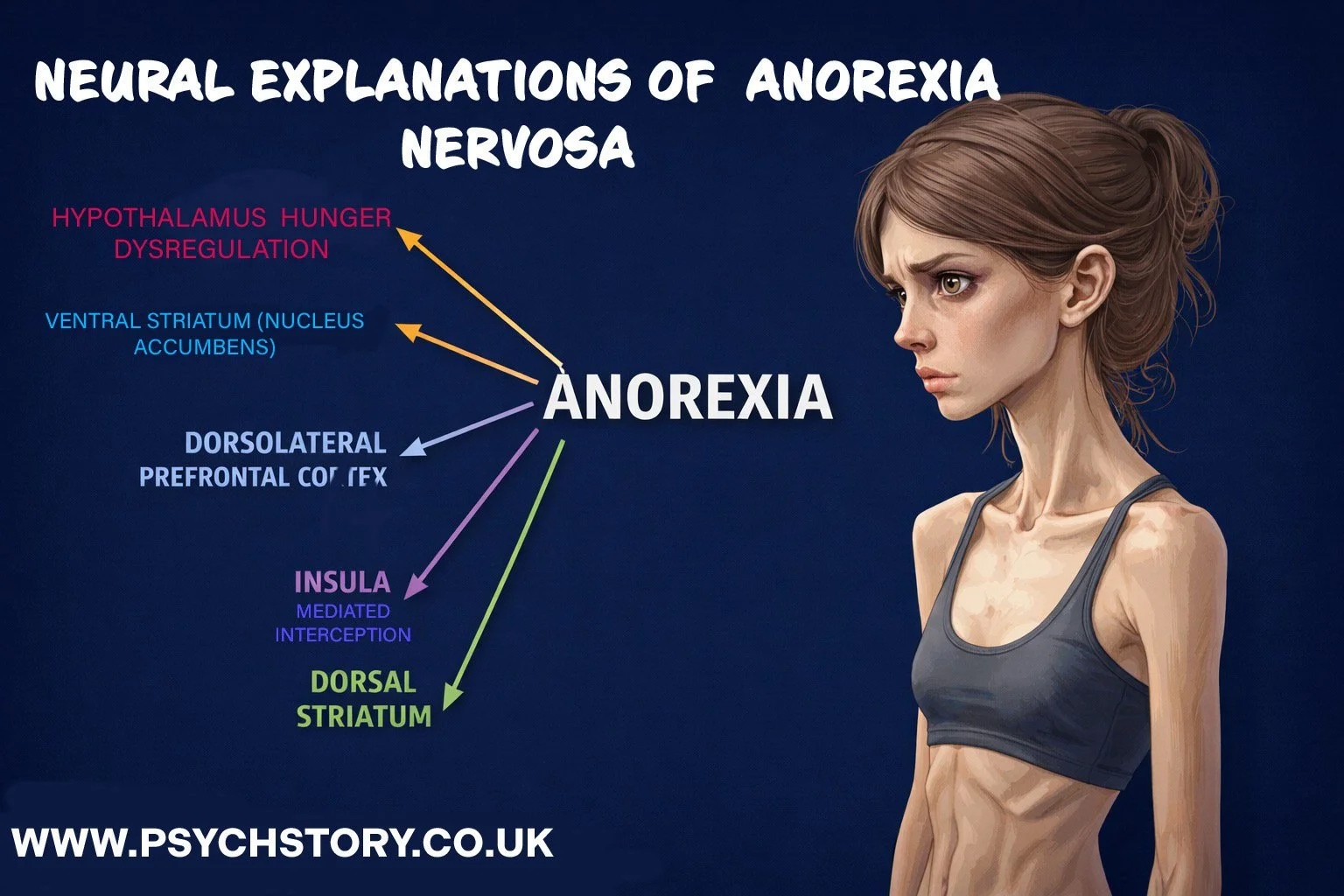
Several interacting neural systems are implicated in anorexia nervosa. The disorder involves abnormalities in circuits that regulate reward, interoception, cognitive control, and homeostatic hunger signalling.
HYPOTHALAMIC ENERGY REGULATION
The hypothalamus regulates hunger and satiety through integrated hormonal and neural signals.
The ARCUATE NUCLEUS contains two key neuronal populations:
• NPY/AGRP NEURONS, which stimulate appetite
• POMC/CART NEURONS, which suppress appetite
In anorexia, signalling within these systems appears dysregulated. Despite severe energy deficit, hunger signalling does not produce normal feeding behaviour. Hormones such as GHRELIN, which normally stimulates appetite, are often elevated, yet food intake remains restricted. This suggests altered hypothalamic responsiveness to hunger signals.
The LATERAL HYPOTHALAMUS, traditionally associated with feeding behaviour, and the VENTROMEDIAL HYPOTHALAMUS, associated with satiety, also contribute to the regulation of energy balance.
REWARD CIRCUITRY AND DOPAMINE
The MESOLIMBIC DOPAMINE PATHWAY plays a central role in reward processing.
Key structures include:
• VENTRAL TEGMENTAL AREA (VTA)
• NUCLEUS ACCUMBENS
• ORBITOFRONTAL CORTEX
In anorexia nervosa, food may produce reduced reward signalling, while behaviours such as dietary restriction or excessive exercise may become reinforcing. Neuroimaging studies have found altered dopamine receptor activity in the striatum, suggesting abnormal reward sensitivity. As a result, behaviours that maintain starvation may become psychologically rewarding.
INSULA AND INTEROCEPTION
The INSULA processes internal bodily states such as hunger, fullness, taste and visceral sensations.
Functional imaging studies have identified altered insula activity in individuals with anorexia nervosa. This may impair INTEROCEPTIVE AWARENESS, meaning individuals may misinterpret or fail to recognise bodily signals related to hunger and satiety. Distorted body perception has also been linked to altered insula functioning.
FRONTAL CORTEX AND COGNITIVE CONTROL
The PREFRONTAL CORTEX, particularly the DORSOLATERAL PREFRONTAL CORTEX, is involved in planning, inhibition and self control.
Individuals with anorexia nervosa often show increased activity in prefrontal control networks when exposed to food related stimuli. This suggests excessive TOP DOWN COGNITIVE CONTROL over eating behaviour, allowing restrictive eating to override normal biological hunger signals.
STRIATAL HABIT CIRCUITS
The DORSAL STRIATUM, which is involved in habit formation and compulsive behaviour, may contribute to the persistence of restrictive eating patterns. Over time, dieting behaviours may become habitual rather than purely goal directed.
SEROTONIN SYSTEM
Altered SEROTONIN (5-HT) signalling has also been observed in anorexia nervosa. Serotonin influences mood, anxiety, impulse control and satiety.
Elevated serotonin activity may contribute to traits commonly associated with anorexia, including ANXIETY, HARM AVOIDANCE AND BEHAVIOURAL INHIBITION.
STRUCTURAL AND FUNCTIONAL BRAIN CHANGES
Neuroimaging studies have identified differences in several brain regions:
• Reduced grey matter volume during prolonged starvation
• Altered activity in the INSULA, ANTERIOR CINGULATE CORTEX AND ORBITOFRONTAL CORTEX
• Abnormal connectivity within reward and cognitive control networks
Some of these changes partially reverse following weight restoration, suggesting that starvation itself can alter brain structure and function.
SUMMARY
Anorexia nervosa involves disruption across multiple neural systems:
• Hypothalamic hunger regulation
• Dopamine based reward processing
• Insula mediated interoception
• Prefrontal cognitive control
• Striatal habit circuitry
• Serotonin related anxiety regulation
The disorder therefore reflects interaction between HOMEOSTATIC FEEDING SYSTEMS, REWARD CIRCUITS AND HIGHER ORDER COGNITIVE CONTROL NETWORKS, rather than a single neural deficit