COMPARATIVE PSYCHOLOGY
ARGUMENTS AGAINST THE USE OF ANIMALS IN RESEARCH (COMPARABILITY)
LIMITED COMPARABILITY OF COGNITION AND EXPERIENCE
Many human psychological functions are either absent or fundamentally different in animals. Complex processes such as language, self-reflection, abstract reasoning, and aspects of emotion and personality are not directly comparable. Even where animals show basic forms of learning or emotion, they cannot report subjective experience, so internal states must be inferred.
Differences also exist at lower levels. Sensory systems vary significantly across species. For example, many animals rely far more on olfaction than vision, or possess specialised sensory mechanisms (e.g. whiskers, lateral lines, echolocation). This means that even basic perceptual experience is not directly equivalent, limiting the validity of generalising findings to humans.
SPECIES DIFFERENCES IN BEHAVIOUR (THEORY-SPECIFIC)
Comparability depends on the behaviour being studied, and in many cases, human behaviour differs qualitatively.
For example, in aggression, most animals display functional, regulated aggression (e.g., for territory or mating), often stopping when the opponent submits. Human aggression, however, can be prolonged, symbolic, and detached from immediate survival goals, including behaviours such as warfare or deliberate cruelty. In eating behaviour, humans are influenced by social, cultural, and cognitive factors, such as dieting, body image, and social norms. Most animals eat in response to biological need and do not show these complex motivations. This limits the extent to which animal models can explain human eating behaviour.
ARTIFICIAL CONDITIONS AND WELFARE CONCERNS
Animal studies are often conducted in controlled laboratory environments that differ substantially from natural conditions. Animals may be caged, isolated, or exposed to stressors, which can produce abnormal behaviour and reduce ecological validity.
In addition, many procedures involve pain, deprivation, or long-term distress, raising ethical concerns about the quality of life experienced by research animals.
DIFFERENCES IN INTELLIGENCE AND BRAIN ORGANISATION
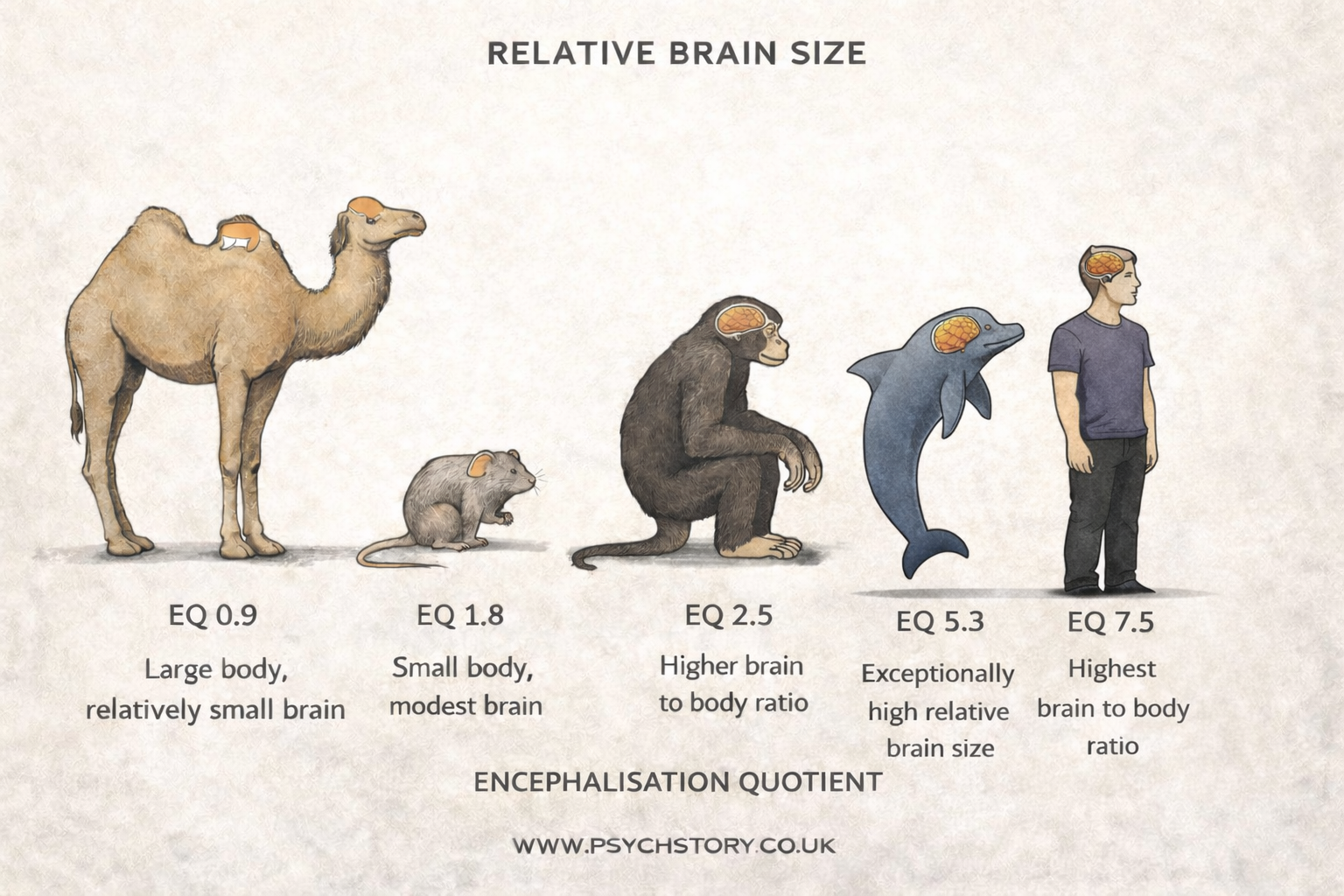
Comparability is further limited by differences in brain development. One measure often cited is the Encephalisation Quotient (EQ), which estimates brain size relative to body size. Humans have a high EQ (approximately 7.0), indicating a disproportionately large brain associated with advanced cognitive abilities.
Many animals have significantly lower EQ values, suggesting more limited cognitive capacity. While EQ is not a complete measure of intelligence, it highlights a gap in cognitive complexity that undermines direct comparisons between animal and human behaviour.
SUMMARY
The central issue is not simply whether animals can model behaviour, but whether they are sufficiently comparable to humans in the specific psychological processes being studied. Differences in cognition, sensory systems, behaviour, and brain structure all limit the validity of generalisation
ENCEPHALISATION QUOTIENT (EQ) AND COMPARABILITY
WHAT IT IS
The Encephalisation Quotient (EQ) is a measure of brain size relative to body size. It compares an animal’s actual brain mass to the expected brain mass for an animal of that size.
An EQ of 1.0 means the brain is about the size expected for that body.
Values above 1.0 indicate a larger-than-expected brain.
Values below 1.0 indicate a smaller-than-expected brain.
Humans have an EQ of approximately 7.0, which is exceptionally high. Most mammals have much lower values.
WHY IS IT RELEVANT TO HUMAN–ANIMAL COMPARABILITY
EQ is used as a rough indicator of cognitive capacity and behavioural complexity. A higher EQ is associated with:
• Greater learning ability
• More flexible problem-solving
• More complex social behaviour
• Greater capacity for memory and planning
Because humans have a significantly higher EQ than most animals, this suggests that human cognition operates at a different level of complexity, particularly in areas such as language, abstract reasoning, self-awareness, and long-term planning.
This limits comparability in psychological research. Even if animals can model basic processes (e.g. conditioning), they may not capture the higher-order cognitive and social processes that shape human behaviour.
FOR EXAMPLE:
• A rat learning through reinforcement does not model human decision-making influenced by beliefs and future goals
• Animal aggression does not fully reflect human aggression shaped by culture, morality, and cognition
LIMITATION OF EQ
EQ is not a perfect measure of intelligence. It does not account for brain organisation, neural connectivity, or specialised adaptations. Some animals with lower EQ still show complex behaviours.
However, it remains useful as a broad indicator of cognitive difference, supporting the argument that animals are not fully comparable to humans in many areas of psychology
ENCEPHALISATION QUOTIENT (EQ): COMPARATIVE VALUES
The following values illustrate differences in brain size relative to body size across species. Humans are a clear outlier, with a substantially higher EQ than other animals.
• Sperm whale — 0.28
• Blue whale — 0.15
• Humpback whale — 0.18
• Bottlenose dolphin — 3.60
• Dolphins (various species) — 2.43–4.45
• Ringed seal — 1.37
• Chimpanzee — 3.00
• Orangutan — 2.65
• Gorillas — 1.5–4.0 (variation across studies)
• Capuchin monkey — 2.52
• Patas monkey — 1.93
• Red colobus — 1.50
• Proboscis monkey — 1.11
• Non-human primates (general range) — 1.04–4.04
• Humans — 7.00
• African elephant — 0.63
• Manatee — 0.32
• Caribou — 0.78
• Wildebeest — 0.68
• Warthog — 0.40
• Hippopotamus — 0.27
• Armadillo — 0.26
• Opossum — 0.39
• Dog — 1.10
• Cat — 1.00
• Horse — 0.86
• Sheep — 0.81
• Mouse — 0.50
• Rat — 0.40
• Rabbit — 0.40
RELEVANCE TO HUMAN–ANIMAL COMPARABILITY
These values demonstrate a clear gradient in cognitive potential. Even the most cognitively advanced animals (e.g. dolphins, chimpanzees) remain substantially below humans. This supports the argument that human cognition operates at a different level of complexity, particularly in areas such as language, abstraction, planning, and self-awareness. As a result, although animals may model basic learning or biological processes, their lower EQ limits the extent to which findings can be generalised to higher-order human behaviour.
CORTICAL FOLDING (GYRIFICATION) AND COMPARABILITY
Cortical folding, or gyrification, refers to the degree of wrinkling of the cerebral cortex, forming gyri (ridges) and sulci (grooves). Greater folding increases the surface area of the cortex without increasing skull size, allowing for more neurons and more complex neural connectivity. Humans have a highly folded cortex, particularly in the prefrontal cortex, which is associated with higher-order functions such as planning, decision-making, abstract reasoning, and social cognition.
Many animals commonly used in research, such as rats and mice, have relatively smooth brains (lissencephalic), with far less cortical folding. This reflects reduced cortical surface area and more limited capacity for complex cognitive processing. Even in animals with more folded brains (e.g., primates or dolphins), the pattern, density, and organisation of folding differ from those in humans, particularly in regions linked to executive function and language.
RELEVANCE TO HUMAN–ANIMAL COMPARABILITY
Differences in cortical folding indicate that brains are not simply scaled versions of one another. The structure and organisation of the cortex differ, not just in size.
This limits comparability because:
• Human cognition relies heavily on highly developed, densely folded cortical regions
• Animals with less folding have reduced capacity for complex integration of information
• Even where behaviour appears similar, the underlying neural architecture may differ
As a result, animal models may be suitable for studying basic processes, but are less valid for explaining higher-level human cognition, where cortical complexity is critical
CORTICAL FOLDING (GYRIFICATION): HIGH VS LOW
HIGH CORTICAL FOLDING (GYRENCEPHALIC BRAINS)
These species have a highly folded cortex, associated with greater cortical surface area and more complex neural processing:
• Humans
• Chimpanzees
• Gorillas
• Orangutans
• Dolphins (e.g. bottlenose dolphin)
• Whales (e.g. sperm whale, humpback whale)
• Elephants
MODERATE CORTICAL FOLDING
• Dogs
• Cats
• Some primates (e.g. capuchin monkeys, macaques)
• Seals
LOW CORTICAL FOLDING (LISSENCEPHALIC OR RELATIVELY SMOOTH BRAINS)
These species have smoother cortices with less surface area and reduced complexity:
• Rats
• Mice
• Rabbits
• Guinea pigs
• Opossums
• Hedgehogs
DIFFERENCES BETWEEN ANIMALS AND HUMANS
CANCER RESEARCH AND FAILED TRANSLATION
Important medical advances have sometimes been delayed, distorted, or misdirected by animal findings that did not translate to humans. Cancer research is one of the clearest examples because animal tumours are usually induced artificially, whereas human cancers arise spontaneously through far more complex genetic, cellular, immune, and environmental interactions. The old claim that 11,600 chemicals have anti-cancer effects in mice but none in humans is not a reliable modern statistic and should not be repeated as a settled fact. The real point is stronger and better supported: a very large proportion of cancer treatments that look promising in animals fail in human trials, and preclinical animal models have been shown to predict human toxicity and efficacy poorly.
SPECIES DIFFERENCES IN DRUG RESPONSE
Animals and humans do not absorb, distribute, metabolise, respond to, or eliminate drugs in the same way. Differences in liver enzymes, receptor sensitivity, immune signalling, and physiology mean that the same substance can produce different, and sometimes opposite, effects across species. This is one reason why toxicology has historically used more than one animal species in the first place: researchers already know that one species does not reliably predict another.
MORPHINE, PENICILLIN, AND CONFLICTING RESULTS
Some of the classic examples are broadly correct, though they need to be stated carefully. Morphine has long had a reputation for causing excitation in cats, even though dose and clinical context matter. Guinea pigs are unusually vulnerable to several antibiotics, including penicillin-class drugs, because these can trigger severe or fatal enterotoxaemia. These examples support the larger argument that one species can tolerate or react to a drug very differently from another.
VITAMIN C AND BIOLOGICAL NON-COMPARABILITY
Mice and rats synthesise their own vitamin C because they retain the enzyme needed to make it, unlike humans. That difference matters because vitamin C is involved in oxidative stress, immunity, and aspects of tumour biology. So, even before an experiment begins, rodent models may have a baseline physiology that is not directly comparable to ours. The old phrase that rodents make “100 times the recommended amount” is too blunt and not the best way to state it. The accurate version is that rodents can produce vitamin C endogenously, whereas humans cannot, and that difference can affect disease modelling.
BOTULINUM, SCOPOLAMINE, AMYLNITRATE, DIGITALIS, AND NOVALGIN
This is the section that most often gets mangled in older anti-vivisection lists. The broad principle is valid: toxins and drugs can affect species very differently. However, several of the dramatic textbook-style examples are either poorly sourced in their exact form or too simplified to present as a hard fact. The botulinum cat-and-mouse example is not secure enough to be repeated literally. The exact scopolamine dose contrast is also not well supported in current reliable sources. The amyl nitrate eye pressure claim, the digitalis dog story, and the Novalgin cat example may contain a kernel of historical truth, but they are difficult to verify confidently from strong modern sources in the simple form in which they are usually quoted. The correct lesson is not that every famous anecdote is perfect. It is that species-specific pharmacology is real, common, and often large enough to make extrapolation uncertain.
THALIDOMIDE AND TGN1412
If recent or well-documented examples are needed, these are much stronger than the older stock anecdotes. Thalidomide remains the classic case of preclinical systems failing to prevent catastrophic human harm, although the slogan that it was fully cleared by modern-style animal testing before release oversimplifies the history. TGN1412 is an even cleaner modern example: it appeared safe in animal studies, including non-human primates, yet caused severe cytokine storm and multi-organ failure in healthy human volunteers. These cases show that animal testing cannot be treated as proof of human safety.
ARTIFICIAL LABORATORY CONDITIONS
Animals in laboratories are handled, confined, restrained, isolated, and exposed to unfamiliar environments. That changes stress hormones, immune function, behaviour, and sometimes even brain chemistry. So the problem is not only a species difference. It is also that animals are often studied under abnormal conditions, which makes their behaviour less representative and weakens extrapolation to humans.
ARTIFICIALLY INDUCED DISEASES
Diseases created in laboratory animals are often artificial approximations of human illness rather than true equivalents. Human diseases usually develop through long-term interactions between genes, age, environment, behaviour, and prior health. By contrast, laboratory disease models are often induced quickly and mechanically. This matters because a treatment that works on an induced model may fail in the real human condition it was supposed to represent. Cancer is again the obvious example.
WHY THIS MATTERS SCIENTIFICALLY
The strongest criticism is not merely ethical. It is scientific. If species differences alter pharmacology, immunity, tumour biology, stress response, and disease progression, then animal data can produce false reassurance, false alarms, and wasted years. A drug may be discarded because it fails in animals even though it might help humans, or it may be advanced because it looks safe in animals even though it is dangerous in people. That is why reviews continue to argue that animal models are often weak predictors of human outcomes.
ALTERNATIVES
This is also why replacement methods are gaining ground. Epidemiology, advanced imaging, human cell and tissue culture, organoids, organ-on-chip systems, and computer-based models are increasingly being developed as more human-relevant alternatives. This is no longer fringe rhetoric. The NIH states that alternative methods hold major promise, and in 2025, the FDA announced plans to phase out animal testing requirements for some drugs when newer human-relevant approaches are available.
CONCLUSION
The defensible conclusion is not that every animal study is worthless. It is that animal-based research cannot be assumed to extrapolate cleanly to humans. Species differences in metabolism, receptor systems, immunity, brain organisation, stress response, and disease processes mean that animal findings can be misleading, non-transferable, or actively counterproductive. That is the real criticism, and it is stronger than the exaggerated versions.
DRUGS TESTED ON ANIMALS THAT STILL CAUSE SERIOUS HUMAN HARM
OPREN (BENOXAPROFEN)
Opren was introduced as a major treatment for arthritis but was withdrawn in 1982 after reports of severe liver toxicity, photosensitivity, and deaths, particularly in older patients. Animal testing did not predict the scale or severity of these adverse effects in humans.
CLIOQUINOL (ENTEROVIOFORM)
Clioquinol was widely used to treat diarrhoea and infections, but became linked to a serious neurological condition known as SMON (subacute myeloid optic neuropathy). This resulted in thousands of cases of paralysis, sensory damage, and visual impairment, particularly in Japan. The drug had appeared acceptable in preclinical testing but caused widespread harm in humans.
THALIDOMIDE
Thalidomide was marketed as safe for pregnant women but caused severe birth defects in over 10,000 children worldwide, alongside miscarriages and infant deaths. It also produced permanent nerve damage in adults. The testing systems in place at the time, including animal studies, failed to detect its teratogenic effects.
ERALDIN (PRACTOLOL)
Eraldin, a beta blocker used to treat heart conditions, was later found to cause a severe syndrome affecting the eyes, skin, and mucous membranes. Patients developed long-term complications, including visual impairment and systemic damage. These effects were not predicted by animal testing.
MODERN EXAMPLE: TGN1412
TGN1412, a monoclonal antibody, passed animal testing, including studies in primates. However, during its first human trial in 2006, it caused a life-threatening immune reaction known as a cytokine storm in healthy volunteers. This highlighted the limits of animal models even in modern, highly regulated research.
WHY THIS MATTERS
These cases demonstrate a consistent problem: animal testing does not reliably predict human outcomes. Species differ in metabolism, immune function, receptor sensitivity, and brain organisation. As a result, drugs that appear safe in animals can still produce severe or unexpected effects in humans.
KEY ISSUE: SPECIES DIFFERENCES
Animals and humans process substances differently. Differences in absorption, distribution, metabolism, and excretion mean that the same drug can have entirely different effects across species. Diseases induced artificially in animals may also fail to replicate the complexity of naturally occurring human conditions.
OVERALL CONCLUSION
Animal research can provide useful biological insight, but it is not a definitive test of safety. These examples show that reliance on animal models alone can delay progress, mislead researchers, and, in some cases, cause significant harm when treatments move into human use
Animal research in psychology raises ethical issues that differ significantly from those involved in human research. While strict guidelines govern the treatment of human participants, historically, animal research has allowed far greater levels of control, intervention, and harm. Procedures such as lesioning, deprivation, and invasive monitoring have been conducted on animals in ways that would be prohibited in human studies.
This creates a central ethical tension. Many areas of psychological and neuroscientific research—particularly those involving brain function, learning, and behaviour—cannot be studied experimentally in humans due to ethical constraints. As a result, researchers have relied on animals to investigate causal relationships, particularly when manipulating the brain or environment is required. Findings are then extrapolated to humans, raising questions about validity and ethics.
KEY ETHICAL ISSUES
The use of animals in research is typically evaluated through several core questions:
Comparability – To what extent are animals sufficiently similar to humans for findings to be generalised? While mammals share biological and neurological similarities, differences in cognition, language, and social structure may limit applicability.
Capacity for suffering – Are animals capable of experiencing pain, distress, or long-term psychological harm? Evidence from neuroscience suggests that many animals, particularly mammals, possess nervous systems capable of pain perception and emotional responses.
Justification of harm – Did the potential scientific or medical benefit justify the level of harm inflicted? This reflects a cost–benefit analysis central to ethical decision-making.
Necessity – Could the research have been conducted using alternative methods, such as human participants, computer modelling, or non-invasive techniques?
Long-term impact – Did the procedure cause lasting harm, including behavioural abnormalities, developmental disruption, or effects on offspring?
Self-awareness and consciousness – Some argue that animals with higher levels of cognition (e.g., primates) may warrant greater ethical protection, given evidence of self-awareness and complex social behaviour.
CLASSIC CONTROVERSIAL STUDIES
Several well-known studies highlight the ethical concerns surrounding animal research:
Harlow and Zimmerman (1959) – Rhesus monkeys were raised in isolation or with surrogate mothers to study attachment. Many developed severe social and emotional disturbances, raising concerns about long-term psychological harm.
Brady (1958) – In the “executive monkey” experiment, monkeys were exposed to stressful conditions involving avoidance learning. Some developed ulcers and died, suggesting extreme physiological stress.
Seligman (1974) – Dogs were exposed to inescapable electric shocks to induce learned helplessness. The study demonstrated important behavioural principles but involved clear distress and suffering.
Riesen (1974) – Sensory deprivation studies in cats and chimpanzees involved restricting visual input during development, leading to permanent perceptual deficits.
Animal research has contributed significantly to understanding areas such as learning, neurobiology, and attachment. However, it raises ongoing ethical concerns about harm, necessity, and justification. The central issue remains whether the scientific value of the research outweighs the moral cost, and whether findings derived from animals can be validly applied to human behaviour.
KEY ETHICAL ISSUES
The use of animals in research is typically evaluated through several core questions:
Comparability – To what extent are animals sufficiently similar to humans for findings to be generalised? While mammals share biological and neurological similarities, differences in cognition, language, and social structure may limit applicability.
Capacity for suffering – Are animals capable of experiencing pain, distress, or long-term psychological harm? Evidence from neuroscience suggests that many animals, particularly mammals, possess nervous systems capable of pain perception and emotional responses.
Justification of harm – Did the potential scientific or medical benefit justify the level of harm inflicted? This reflects a cost–benefit analysis central to ethical decision-making.
Necessity – Could the research have been conducted using alternative methods, such as human participants, computer modelling, or non-invasive techniques?
Long-term impact – Did the procedure cause lasting harm, including behavioural abnormalities, developmental disruption, or effects on offspring?
Self-awareness and consciousness – Some argue that animals with higher levels of cognition (e.g. primates) may warrant greater ethical protection due to evidence of self-awareness and complex social behaviour
ANIMAL ETHICS: COST–BENEFIT APPROACH
Despite the introduction of stricter ethical guidelines, it remains permissible to cause harm to animals in research when the purpose of the study is judged sufficiently valuable. In the UK and across much of Europe, this is regulated through a formal harm–benefit analysis, in which the potential suffering, injury, or death of animals is weighed against the expected scientific or medical gains.
Many animals, particularly mammals, possess nociceptive systems comparable to those of humans, including similar pain receptors and neural pathways. This means they are capable of experiencing not only physical pain but also forms of distress, making their use in research ethically contentious.
The regulatory framework does not eliminate harm but seeks to justify and minimise it. Research is permitted only when there is no viable alternative and when the anticipated benefits—such as advances in knowledge or the development of medical treatments—are considered to outweigh the ethical costs. This reflects a fundamentally utilitarian position, where animal suffering can be sanctioned if it is believed to produce greater overall benefit.
ARGUMENT FOR USING ANIMALS IN RESEARCH
UNDERSTANDING HOW THE BRAIN WORKS
A substantial proportion of knowledge about brain structure and function has come from invasive animal research. Lesion studies, in which specific areas of an animal’s brain are damaged, and behaviour is observed, have been central to identifying the functions of different brain regions. This approach contributed to early localisation findings and continues to inform modern neuroscience, particularly where direct experimentation on humans would be unethical.
ETHICAL JUSTIFICATION AND PRACTICAL NECESSITY
Many areas of research cannot be conducted on humans due to ethical constraints. Studies involving deprivation, invasive procedures, or induction of pathology would be unacceptable in human participants. Animal models, therefore, allow investigation of conditions such as aggression, obesity, addiction, and schizophrenia, where understanding underlying mechanisms has clear social and clinical value. The justification is typically utilitarian, weighing potential harm to animals against benefits to human health and knowledge.
CHEMICAL BASIS OF BEHAVIOUR
Animal studies have been critical in identifying the role of neurotransmitters and hormones. Pharmacological manipulation, administering drugs to alter brain chemistry, has allowed researchers to examine how systems such as dopamine, serotonin, and cortisol influence behaviour. Much of what is known about addiction, mood disorders, and reward pathways originates from this work. At the same time, species differences in drug response remain a recognised limitation.
ELECTRICAL ACTIVITY AND NEURAL FUNCTION
Electrical stimulation studies in animals have provided direct evidence of how neurons and neural circuits operate. By stimulating specific brain regions and observing resulting behaviours, researchers have mapped functional areas and clarified how signals are transmitted within the nervous system. This work underpins modern techniques such as deep-brain stimulation used in humans.
MEDICAL ADVANCES AND DISEASE TREATMENT
Animal research has played a role in the development of vaccines and treatments for major diseases, including tuberculosis, polio, measles, mumps, rubella, and tetanus, as well as in understanding diabetes. Surgical techniques such as organ transplantation and open-heart surgery were refined through animal models before being applied to humans.
BIOLOGICAL SIMILARITIES BETWEEN SPECIES
Supporters of animal research emphasise the significant biological continuity across species. Humans share a large proportion of genes with other organisms, including approximately 98 per cent with chimpanzees and a substantial proportion even with simpler organisms such as fruit flies. Mammalian brains share common structural features, including the organisation of the forebrain, midbrain, and hindbrain, as well as similar endocrine and nervous systems.
SHARED REWARD AND ADDICTION SYSTEMS
The mesolimbic dopamine system, central to reward and motivation, is highly conserved across mammals. Because animals and humans share this pathway and many of the genes that regulate it, animals can model processes such as reinforcement, addiction, and compulsive behaviour. This has made them particularly useful in addiction research.
NEUROBIOLOGICAL SIMILARITY IN RODENTS
Rodents, commonly used in laboratory research, show strong similarities to humans at the neurochemical level. They use the same major neurotransmitters, including dopamine, serotonin, and glutamate, and share similar receptor systems and synaptic mechanisms. Proteins involved in synaptic transmission and neural signalling are also highly conserved, allowing findings to be translated, with caution, to human brain function.
OVERALL EVALUATION
Animal research has contributed significantly to neuroscience and medicine, particularly in areas where human experimentation is not possible. Its value rests on biological similarities between species and the ability to conduct controlled, invasive studies. However, its limitations remain tied to species differences, especially in higher cognitive processes and drug responses, requiring careful interpretation of findings
GENETIC SIMILARITIES BETWEEN HUMANS AND ANIMALS
CHIMPANZEE (PAN TROGLODYTES)
Chimpanzees share approximately 98–99% similarity with human DNA. Both species have around 20,000–25,000 genes, with many protein-coding genes either identical or extremely similar. Only a small number of genes appear to be uniquely human. This high overlap underpins their relevance in research, although ethical constraints now limit their use.
BONOBO (PAN PANISCUS)
Bonobos are as genetically similar to humans as chimpanzees, with around 98–99% DNA similarity. Differences between humans, chimpanzees, and bonobos are less about the presence of genes and more about how genes are regulated and expressed. Behavioural differences, therefore, arise from gene expression patterns rather than entirely different genetic content.
MOUSE (MUS MUSCULUS)
Mice have approximately 20,000–25,000 genes, similar to humans. Around 85% of mouse genes have a human homolog. Core biological systems, including neural signalling and metabolism, are highly conserved, which explains their widespread use in genetic and biomedical research.
ZEBRAFISH (DANIO RERIO)
Zebrafish possess around 25,000 genes. Approximately 70% of human genes have a zebrafish equivalent, and many genes linked to human disease are present. Their transparent embryos allow direct observation of gene function, making them useful for developmental and genetic studies.
FRUIT FLY (DROSOPHILA MELANOGASTER)
Fruit flies have around 14,000 genes, far fewer than humans, yet approximately 60% of human genes have a recognisable counterpart. Around 75% of genes involved in human diseases are represented in some form. Despite their simplicity, they are extensively used in genetic research because fundamental cellular and developmental processes are conserved.
DOLPHIN (E.G. BOTTLENOSE DOLPHIN, TURSIOPS TRUNCATUS)
Dolphins show high genetic similarity to humans, with roughly 80–90% of genes having human equivalents. They also have large, highly folded brains and complex social behaviour. However, differences in brain organisation and ecology limit direct comparison despite genetic overlap.
HUMAN GENETIC VARIATION (WITHIN SPECIES)
Humans are genetically extremely similar to one another. Any two humans share approximately 99.9% of their DNA. The remaining 0.1% accounts for all individual differences, including physical traits, disease susceptibility, and aspects of behaviour. This highlights that even very small genetic differences can produce significant variation, which is relevant when comparing across species.
WHY THIS MATTERS
Across species, gene number is broadly similar, and many genes are shared. What differs is gene regulation, expression, and interaction within complex systems. This means animal research can model basic biological processes, but differences in expression, brain organisation, and environment limit direct extrapolation to human behaviour